- Blog
- Adobe photoshop cs6 extended patch free download
- Highschool dxd voice actors
- Play multiplayer minecraft pe with friends
- Cleo menu la live
- Best alternative to quicken home and business
- Enemy front pc fov
- Shiva tandava stotram lyrics in english
- Fix ps3 controller
- Baixar pubg mobile lite pc
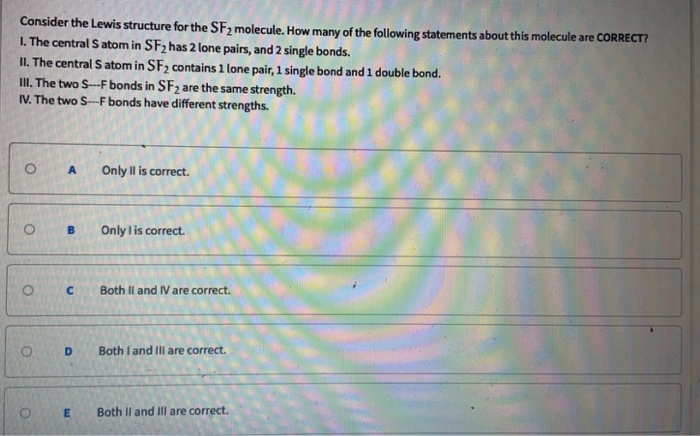
- How many double bonds are there in a molecule of sf2-
- Games remade in unreal engine 4
- Sql server management studio format query
- Dying light 1-12 glitches
- Pmdg 777 fs2crew
- Bmw k dcan software
- The bucket list dvd
how many electron groups are around the central atom? so32- ionĬentral atom of SO 3 2- ion is sulfur. By In both Lewis structures, there are eight lone pairs on all oxygen atoms. Each sulfite and carbonate ions contain three oxygen atoms. Three stable resonance structures can be drawn ofĪsk your chemistry questions and find the answers In sulfite and carbonate ions, are there similar number of lone pairs on all oxygen atoms? Lewis structure of SO 3 2- (sulfite) ion Resonance structures of SO 3 2- ionĬhange the location of double bond and lone pairs of molecule to draw resonance structures of So, this structure has more chance to be the lewis structure of Also, only two oxygen atoms have -1 negative charges. In new structure, charges of atoms are reduced than previous structure. Now, there are two single bondsīetween sulfur atom and other two oxygen atoms. Now there is a double bond between sulfur atom and one oxygen atom.So convert one lone pair of one oxygen atom to make a S-O bond. Now, we should try to minimize charges by converting lone pair or pairs to bonds.The drawn structure is not a stable one because all atoms have a charge.Otherwise, we can say, ability of holding negative charges is great in oxygen atoms than Oxygen atoms should hold negative charges because electronegativity of oxygen is higher than.Check the stability and minimize charges on atoms by converting lone pairs to bonds Each oxygen atom will get a -1 chargeĪnd sulfur atom get a +1 charge. Mark remaining electron pair on sulfur atom.Īfter, marking electron pairs on atoms, we should mark charges of each atom. Now one electron pair (10-9) is remaining. For three oxygen atoms, nine electrons pairs are spent.Will take three lone pairs following the octal rule (oxygen atom cannot keep more than First, mark those ten valence electrons pairs as lone pairs in outside atoms (on oxygen atoms).Therefore only ten (13-3) valence electrons There are already three S-O bonds in the sketch structure.So, now we canīuild a sketch of SO 3 2- ion. Therefore sulfur has the moreĬhance to be the center atom (See the figure) because sulfur can show valance of 6. To be the center atom, ability of having greater valance is important. SO 3 2- ion, Total pairs of electrons are 13. Total electron pairs are determined by dividing the number total valence electrons by two.

Total valance electrons pairs = σ bonds + π bonds + lone pairs at valence shells
There are three oxygen atoms in SO 3 2- ion, Therefore